Galectin-3, Sepsis, and the Breakthrough Device That Could Change Critical Care Forever
A Web Blog Article | Science & Medical Innovation
Every two seconds, somewhere in the world, someone dies from sepsis. That is not a typo. Sepsis, a runaway inflammatory response to infection, kills more people each year than breast cancer, prostate cancer, and HIV combined. Yet most people have barely heard of it. What is even more remarkable is that for decades, scientists have been hunting for a single molecular villain at the heart of this catastrophe. They may have finally found it, a tiny protein called Galectin-3, and a new medical device is now being engineered to rip it right out of your blood.

This article takes you deep into the biochemistry, the clinical realities of sepsis, and the story of how one molecule became the focus of a potential revolution in critical care medicine.
What Is Sepsis And Why Should You Care?
Let’s start with definitions, because these terms are often used interchangeably but mean very different things:
Bacteremia is simply the presence of bacteria in the bloodstream. Think of it like a few unwanted guests slipping through the front door of your body. They are detectable but have not yet caused chaos.
Septicemia is an older, largely deprecated term that described active bacterial infection of the bloodstream, essentially bacteremia that had become a serious clinical illness.
Sepsis is something entirely different and far more dangerous. Sepsis is not the infection itself, it is your body’s own catastrophically dysregulated immune response to an infection. It is the fire your immune system lights to fight the enemy that burns your own house down.
Clinically, sepsis is diagnosed using a framework called the Sepsis-3 criteria, established in 2016. According to these criteria, sepsis occurs when an infection leads to life-threatening organ dysfunction, characterized by an increase of 2 or more points on the SOFA score (Sequential Organ Failure Assessment), which measures the failure of respiratory, coagulation, liver, cardiovascular, nervous, and renal systems.
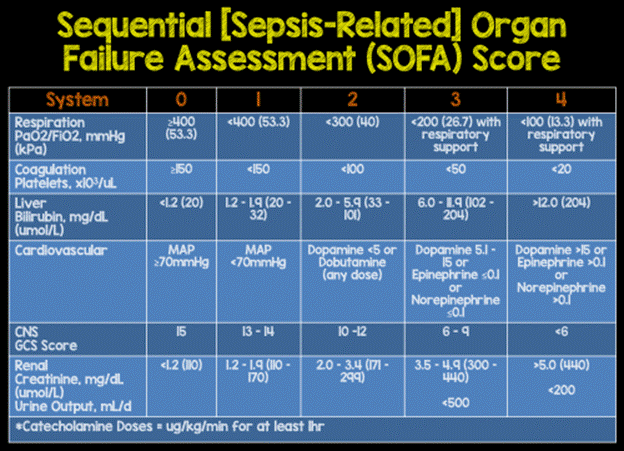
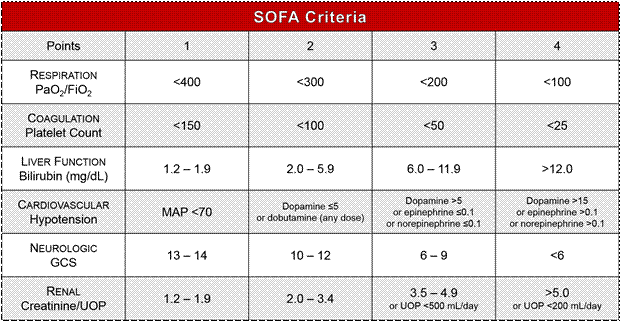
The progression beyond sepsis is called septic shock, defined by the need for vasopressor medications to maintain blood pressure and elevated blood lactate levels, indicating the body’s cells are suffocating for energy.

Globally, sepsis is responsible for an estimated 11 million deaths per year, accounting for approximately 19% of all global deaths. In the United States alone, nearly 1.7 million adults develop sepsis annually, and roughly 270,000 die from it. The economic burden on the U.S. healthcare system exceeds $62 billion per year.
Who or What Causes Sepsis?
Sepsis is not caused by one pathogen. It can be triggered by a shockingly broad array of microorganisms:
Common bacterial culprits include Escherichia coli (E. coli), Staphylococcus aureus (including the dreaded antibiotic-resistant MRSA), Streptococcus pneumoniae, Klebsiella pneumoniae, and Pseudomonas aeruginosa. These are the usual suspects, organisms well-known to emergency physicians and ICU teams.
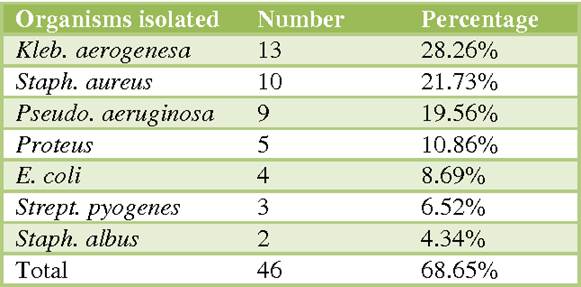
Less common but equally dangerous are organisms like Listeria monocytogenes, Neisseria meningitidis (meningococcus), Clostridium species, Acinetobacter baumannii, and Enterococcus faecalis. Some of these are notorious for antibiotic resistance, making treatment nightmarishly complex.
Fungal sepsis, though less frequent, carries an alarmingly high mortality rate. Candida species (Candida albicans, Candida glabrata) and Aspergillus fumigatus are among the primary fungal offenders, particularly in immunocompromised patients — those on chemotherapy, organ transplant recipients, or individuals with advanced HIV/AIDS.
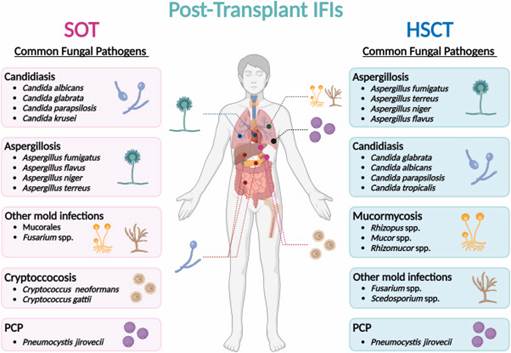
Viral sepsis gained global attention with SARS–CoV-2 (COVID-19), but influenza, hantavirus, Ebola, and herpes simplex virus can all trigger a septic-like immune cascade. The COVID-19 pandemic gave the world an unwanted master class in what cytokine storms, a hallmark of sepsis, look like at epidemic scale.
The Terrible Complications: When Sepsis Attacks the Body
Sepsis doesn’t just cause fever and low blood pressure. It launches a systemic assault on virtually every organ system, producing complications that read like a medical horror story:
Disseminated Intravascular Coagulation (DIC) is among the most feared. The inflammatory cascade activates clotting pathways throughout the entire bloodstream simultaneously. Tiny clots, emboli, form in small blood vessels across organs, cutting off their oxygen supply. This can cause fingers, toes, and even limbs to die from ischemia, sometimes requiring amputation. Paradoxically, all these clotting factors get used up, and the patient then begins to bleed uncontrollably.
Septic emboli can seed heart valves, causing infective endocarditis, a condition in which bacteria or fungi colonize the delicate valve leaflets, forming destructive vegetations that can destroy valve architecture, cause the valve to leak or obstruct, and embolize clots to the brain, kidneys, and other organs.
Vascular complications include septic thrombosis in deep veins, mycotic aneurysms (weakening of arterial walls due to infection), and peripheral arterial occlusion. The systemic vasodilation that occurs in sepsis causes blood to pool inappropriately, crashing blood pressure and starving vital organs of perfusion.
Acute Respiratory Distress Syndrome (ARDS), acute kidney injury, liver failure, encephalopathy (brain dysfunction), and adrenal insufficiency round out the devastating catalog of sepsis complications.
Treating the Unknown Enemy: Empiric Therapy in Sepsis
Here is one of the most challenging aspects of sepsis management: when a patient arrives critically ill, you often don’t know what pathogen is causing it. Blood cultures take 24–72 hours to grow. Every hour of delay in starting antibiotics increases mortality by approximately 7–10%. So, physicians must act fast before the identity of the pathogen is confirmed.
This is called empiric antibiotic therapy, a calculated, educated bet based on the likely source of infection, the patient’s history, and local patterns of antibiotic resistance. The goal is broad-spectrum coverage: cast the net wide and catch the most likely offenders.
Common empiric antibiotic regimens include:
Piperacillin-tazobactam (Zosyn): a broad-spectrum penicillin-beta-lactamase inhibitor combination covering gram-negative rods, gram-positive organisms, and anaerobes.
Vancomycin: the gold standard for gram-positive coverage, particularly MRSA.
Meropenem or imipenem (carbapenems): “last-resort” broad-spectrum agents for highly resistant organisms or polymicrobial infections.
Cefepime or ceftriaxone: broad-spectrum cephalosporins for community-acquired or hospital-acquired infections.
Micafungin or fluconazole: added empirically when fungal sepsis is suspected, especially in immunocompromised patients.
Supportive care is equally critical: intravenous fluids to restore blood pressure, vasopressors (like norepinephrine) when fluids aren’t enough, supplemental oxygen and mechanical ventilation for respiratory failure, and dialysis for acute kidney injury. The “Surviving Sepsis Campaign” is an international initiative that has codified these interventions into evidence-based bundles of care, and adherence to these protocols measurably saves lives.
Despite all these tools, the fundamental problem remains: current treatments manage symptoms and fight the infection, but they do not address the underlying biological derangement — the molecular firestorm — that is actually killing the patient.
Enter Galectin-3: The Molecular Arsonist
Now we arrive at the biochemistry, the molecular heart of the story.
Galectin-3 (Gal-3) belongs to a family of proteins called lectins, which proteins that bind to specific sugar structures (carbohydrates) on the surfaces of cells and molecules. Think of lectins as the body’s molecular reading glasses: they scan the surface of cells, recognizing specific carbohydrate “words” and triggering biological responses accordingly.
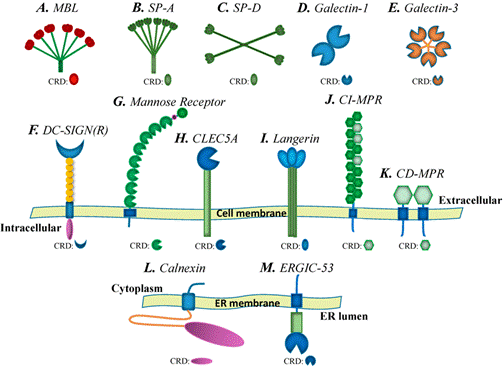
Lectin Structure
Gal-3 was first identified and characterized in the 1980s. Researchers initially discovered it as a protein expressed on the surface of macrophages, the large immune cells that patrol the body eating cellular debris and pathogens. Early research identified it under several names: Mac-2 antigen, CBP35, L-29, and others, reflecting its discovery by multiple independent research groups before it was recognized as one and the same protein.
Structurally, Galectin-3 is unique among the galectin family. While most galectins have either one or two carbohydrate-recognition domains (CRDs), the sugar-binding “hands” of the protein, Galectin-3 has only one CRD, but it also possesses a long, flexible N-terminal domain. This tail allows multiple Galectin-3 molecules to self-assemble into oligomers, clusters of five or more molecules that form a lattice-like cage. This pentameric structure can crosslink multiple carbohydrate-bearing molecules simultaneously, effectively “stapling” cells, receptors, and signaling molecules together and amplifying inflammatory signals in a powerful, dangerous way.
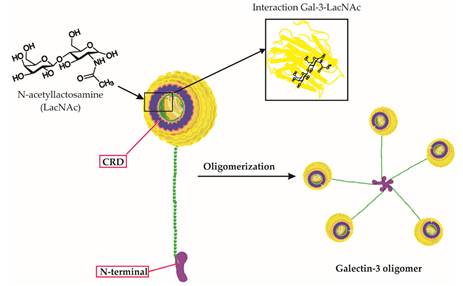
At the molecular level, Gal-3 binds to N-acetyllactosamine (LacNAc) disaccharide units, sugar structures found abundantly on the surface of immune cells, endothelial cells (cells lining blood vessels), and extracellular matrix components. When Gal-3 crosslinks these structures, it initiates intracellular signaling cascades involving NF-κB (nuclear factor kappa-light-chain-enhancer of activated B cells), a master regulator of inflammation. This triggers the release of pro-inflammatory cytokines including TNF-α, IL-1β, IL-6, and IL-8, the very molecules that drive the cytokine storm of sepsis.
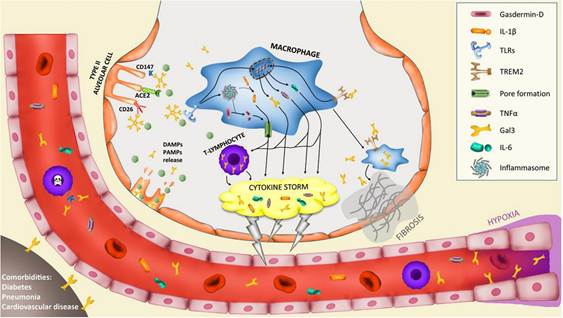
In healthy physiology, Gal-3 plays constructive roles: it participates in wound healing, promotes phagocytosis (the eating of pathogens by immune cells), and helps regulate cell death. It is, in small amounts, a useful biological tool. But in serious infection, Gal-3 levels spike dramatically in the bloodstream. At high concentrations, it acts less like a tool and more like a match dropped in a gasoline-soaked room.
Specifically in sepsis, elevated circulating Gal-3 promotes uncontrolled macrophage activation, drives fibrosis in injured organs, amplifies neutrophil-mediated tissue damage, promotes the pro-coagulant state that leads to DIC and organ-destroying microemboli, and critically, impairs the body’s ability to resolve inflammation. It becomes, in essence, a molecular brake failure, preventing the immune system from switching off even after the infection is controlled.
Research has further established that Gal-3 is elevated in heart failure, acute kidney injury, liver fibrosis, and cancer, confirming its role as a central driver of inflammatory and fibrotic disease across multiple organ systems.
XGal-3: Engineering a Solution to a Molecular Problem
So if elevated Galectin-3 is a key driver of the sepsis catastrophe, what if you could simply remove it from the blood?
That is the elegantly straightforward premise of XGal-3®, a medical device developed by Eliaz Therapeutics, founded by Dr. Isaac Eliaz, a physician and researcher who has studied Galectin-3 for over three decades and whose own personal loss to immune dysregulation fueled a lifelong mission to combat it.
XGal-3® works on a principle similar to dialysis, but with a critical molecular twist. In standard dialysis, a patient’s blood is passed through a machine that filters out waste products using a semi-permeable membrane. XGal-3® uses therapeutic apheresis, a well-established clinical procedure, but instead of a generic membrane, the blood plasma passes through a column containing proprietary anti-Galectin-3 ligand traps: specially engineered molecules that selectively capture and remove Gal-3 from circulation. Think of it as a highly specific molecular fishing net, designed to catch only Galectin-3 while leaving everything else alone.
The device is designed to attach to existing apheresis machines, specifically the Terumo Spectra Optia® platform, already in widespread use in hospitals globally — making it potentially deployable without massive infrastructure investment.
What makes XGal-3® particularly remarkable from a scientific standpoint is that it removes both free and bound Gal-3 from circulation. In sepsis, Gal-3 exists in multiple forms in the bloodstream, free-floating molecules and molecules already crosslinked to cell-surface glycoproteins. The device’s engineered ligand traps capture both forms, offering a more comprehensive Gal-3 depletion than pharmacologic inhibitors, which typically only block the protein’s binding activity.
The early results are striking. In large animal studies, XGal-3® demonstrated the ability to halt the sepsis cascade within hours, a benchmark no currently approved treatment has achieved. Importantly, no adverse side effects were observed in these studies, a critical safety milestone. No serious complications have emerged in preclinical work, supported by two NIH grants totaling approximately $1.7 million and protected by over 60 patents across 34 countries.
The FDA has awarded XGal-3® its Breakthrough Device Designation, a status reserved for technologies that show promise for treating serious or life-threatening conditions where no adequate alternatives exist. This designation accelerates regulatory engagement and can shorten the path to market, giving critically ill patients potential access to this technology sooner.
Eliaz Therapeutics is now advancing toward investigator-led clinical trials in ICU settings at top academic medical centers, with the goal of demonstrating proof-of-concept in humans and building the evidence base needed for FDA approval.
The Bottom Line
Sepsis is a global killer that has eluded effective treatment because it has been approached primarily as an infection problem rather than an immune dysregulation problem. Galectin-3, a protein whose dangerous complexity is written into its very molecular architecture,appears to be one of the central conductors of the inflammatory orchestra that kills sepsis patients, even after the infection itself is controlled.
XGal-3® represents a conceptual leap: instead of drugging the immune system or fighting pathogens, it proposes removing a specific molecular instigator directly from the bloodstream. It is a marriage of extracorporeal medicine and precision molecular biology; dialysis meets targeted therapy.
Whether XGal-3® will fulfill its extraordinary promise will be answered in the clinical trials ahead. But what is already clear is this: the era of targeting the molecular roots of sepsis has arrived. And for the millions of patients who enter ICUs every year teetering on the edge of a storm they cannot see that may mean everything.
Leave a comment