What pain management patients — and opioid-therapy advocates — need to know about buprenorphine’s hidden cost to oral health
Suboxone has become a contentious fixture of American medicine. Marketed primarily for opioid use disorder but increasingly pushed onto chronic pain patients, it carries a side effect that regulators were slow to acknowledge and many prescribers still downplay: rapid, severe dental decay. In 2022 the FDA added a warning to oral buprenorphine products after cataloging hundreds of cases of tooth loss, fractures, and cavities in patients with previously healthy mouths. This isn’t a hygiene failure; it’s chemistry, and it deserves a seat at the table alongside efficacy, withdrawal, and quality-of-life when weighing buprenorphine against a full-agonist opioid.
What Suboxone Actually Is
Suboxone combines buprenorphine with naloxone in a 4:1 ratio. Buprenorphine is the active agent; naloxone is an abuse deterrent that is poorly absorbed orally but blocks opioid receptors if the product is injected. Buprenorphine itself is available in several forms:
- Sublingual films (Suboxone, Zubsolv) and tablets (Subutex, generic)
- Buccal films for pain (Belbuca)
- Transdermal patches for pain (Butrans)
- Subcutaneous extended-release injection (Sublocade)
The dental risk is tied entirely to the oral formulations. The patch and injection bypass the oral cavity and show no comparable dental signal, which tells you the mechanism is local, not systemic.
Structure and Receptor Pharmacology
Buprenorphine is a semi-synthetic derivative of thebaine, an opium alkaloid. Structurally it retains the core morphinan scaffold shared by morphine, oxycodone, and hydrocodone — the rigid pentacyclic system with oxygen bridge, tertiary amine, and aromatic A-ring that defines classical mu agonists. Two modifications change everything: a bulky tert-butyl side chain at C7 and a cyclopropylmethyl group on the nitrogen. These increase lipophilicity dramatically and produce extraordinarily tight mu-receptor binding, with a dissociation half-life measured in hours.
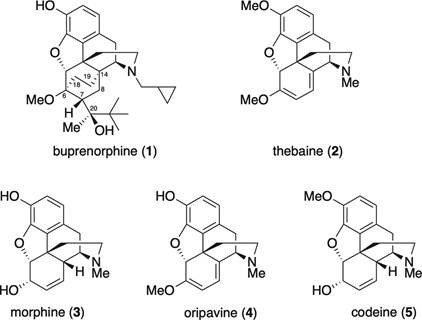
Buprenorphine structure and comparisons
Buprenorphine docks into the same orthosteric pocket of the mu-opioid receptor that morphine does, contacting the same key residues (Asp147, Tyr148, His297). That homology is why it relieves pain and suppresses withdrawal.
The mu receptor is a class A G-protein coupled receptor. When a full agonist like morphine binds, two parallel intracellular events unfold. First, the Gαi/o subunit dissociates and inhibits adenylyl cyclase, dropping cyclic AMP, while Gβγ opens GIRK potassium channels and closes voltage-gated calcium channels, hyperpolarizing neurons and blunting neurotransmitter release. That is the analgesic arm. Second, the receptor is phosphorylated by GRK kinases and recruits beta-arrestin-2, which drives internalization, desensitization, and much of the respiratory depression, constipation, and tolerance attributed to opioids.
Buprenorphine is a partial agonist at mu: higher affinity than morphine but only partial G-protein activation, producing the ceiling effect on respiratory depression. It is also a biased ligand, recruiting beta-arrestin-2 poorly, and it antagonizes the kappa receptor while partially engaging the nociceptin receptor. Pharmacologically elegant. Dentally, irrelevant.
The Dental Damage: A Chemical, Not Behavioral, Problem
Tooth enamel is hydroxyapatite — Ca₁₀(PO₄)₆(OH)₂ — a crystalline mineral that dissolves whenever the pH at its surface drops below 5.5. The mouth is normally buffered near neutral by saliva, which is supersaturated with calcium and phosphate and rich in bicarbonate. Every acid challenge from food or drink is neutralized within minutes, and minerals are redeposited. This remineralization is what keeps teeth intact across a lifetime.
Suboxone disables the system in four simultaneous ways.
1. The drug itself is acidic
Buprenorphine films and tablets are formulated at low pH for solubility and stability. Dissolved in saliva, they drop the local pH to 3.5–4.5, which is well below the critical threshold. Hydrogen ions penetrate the hydroxyapatite lattice, protonate phosphate groups, and liberate calcium into saliva. This is straight acid etching, the same chemistry used to dissolve limestone.
2. Contact time is prolonged
A film takes 10 to 30 minutes to dissolve, and patients are told not to rinse or swallow excessively during absorption. The acid bath sits against enamel far longer than anything else people put in their mouths.
3. Saliva production crashes
Mu-opioid receptors in the brainstem salivary nuclei and on parasympathetic fibers to the salivary glands suppress acetylcholine release when activated, cutting salivary flow by 50 to 75 percent in chronic users. No saliva means no bicarbonate buffer, no calcium-phosphate reservoir, and no mechanical clearance. The remineralization arm of the equation is amputated.
4. The microbiome shifts and the pellicle thins
Dry, acidic mouths favor aciduric bacteria like Lactobacillus, which ferment residual carbohydrate through glycolysis to lactic acid (pKa 3.86). Meanwhile, amphipathic buprenorphine molecules adsorb onto enamel and displace the salivary proteins (statherin, mucins, proline-rich proteins) that form the protective acquired pellicle. The barrier thins, and protons penetrate more easily.
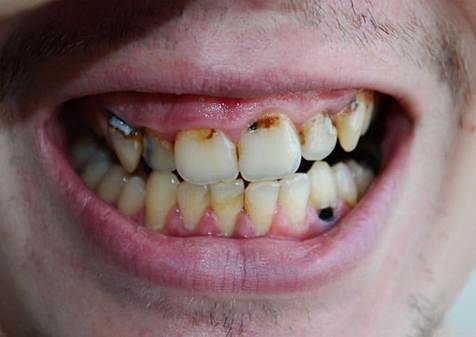
The cumulative effect is catastrophic. Reports describe patients losing multiple teeth within one to two years of starting Suboxone, with decay at unusual sites — cervical margins, incisal edges, between teeth — rather than the usual occlusal pits. These patterns are pathognomonic for chemical erosion, not ordinary bacterial caries.
How Suboxone Compares to Other Opioids
Advocates for traditional opioid therapy often ask whether their preferred medications cause the same damage. The answer is largely no — and the reasons are instructive.
| Factor | Suboxone (sublingual) | Oral full-agonist opioids |
| Oral contact time | 10–30 minutes of dissolution | Seconds; swallowed whole |
| Local pH at tooth surface | 3.5–4.5 | Near neutral |
| Dry mouth effect | Strong, chronic | Present but typically milder |
| FDA dental warning | Yes (2022) | No equivalent class warning |
Oral morphine, oxycodone, and hydrocodone all cause some xerostomia through the same central mechanism, and long-term opioid users do show elevated caries rates. But without the direct acid bath and prolonged mucosal contact, damage is slower, more diffuse, and more responsive to ordinary hygiene. Methadone liquid — often sweetened and sipped slowly — produces a pattern closer to Suboxone. Transdermal buprenorphine and the Sublocade injection avoid the oral hit entirely and show no comparable dental signal.
Prevention: What Actually Works
If you are on sublingual buprenorphine and intend to stay on it, the biochemistry dictates the countermeasures: shorten acid exposure and restart remineralization as quickly as possible after each dose.
- Do not rinse, brush, or drink immediately before dosing. You need saliva present to dissolve the drug.
- After the film or tablet is fully dissolved, wait a few minutes, then rinse with plain water or a diluted baking soda solution (½ teaspoon in a cup of water). Bicarbonate neutralizes residual acid and raises pH above the critical threshold.
- Do not brush for at least 30 minutes afterward. Softened enamel is physically removed by abrasive brushing at that moment.
- Use a prescription 5,000 ppm fluoride toothpaste at bedtime. Fluoride substitutes into the lattice as fluorapatite, which resists dissolution to pH 4.5.
- Add a calcium-phosphate remineralizing agent (CPP-ACP, sold as MI Paste). It replaces the ions the drug strips out.
- Treat dry mouth aggressively. Xylitol gum stimulates saliva and inhibits S. mutans; pilocarpine or cevimeline can be prescribed for severe cases.
- See a dentist every three to four months, not every six. Early white-spot lesions are reversible; cavitated lesions are not.
- Discuss non-oral formulations with your prescriber. The Sublocade injection and Butrans patch deliver buprenorphine without touching enamel.
The Bottom Line
The case against Suboxone’s dental profile isn’t a talking point. It is chemistry you can measure with pH paper. A partial mu agonist with fascinating receptor-level biology has been delivered in a vehicle that bathes teeth in acid for half an hour at a time while simultaneously switching off the body’s repair system. That design choice was made for pharmacokinetic convenience, not patient welfare, and the consequences are visible in thousands of mouths.
For pain patients considering their options, and for advocates arguing the pendulum has swung too far toward buprenorphine and away from traditional opioid therapy, this is a legitimate piece of the argument. Informed consent means knowing that the price of the ceiling effect on respiratory depression may, for some people, be their teeth, and that cheap countermeasures exist if you start using them from day one. Talk to your dentist before your next refill.
Leave a comment