A Quiet Discovery Out of Germany Could Change Everything We Know About Osteoporosis
For decades, the conversation around osteoporosis treatment has felt a bit like a losing battle. You take calcium. You get enough vitamin D. You lift weights. You hope for the best. Current medications can slow bone loss, but many come with serious long-term side effects and none of them truly rebuild bone from the inside out the way your body naturally would in its prime.
That may be about to change.
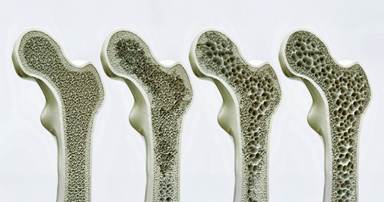
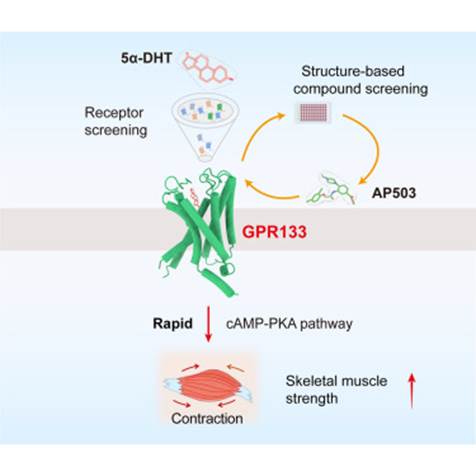
Researchers at Leipzig University in Germany have identified a little-known molecular “switch” on bone cells, a receptor called GPR133 that, when activated, can dramatically boost bone formation, reverse osteoporosis-like bone loss, and even amplify the benefits of exercise. The compound they used to flip that switch? A molecule called AP503. And the results in mice have been striking enough to turn heads across the scientific community.
Who Made This Discovery — and Where?
The work was led by Professor Ines Liebscher, MD, PhD, a researcher at the Rudolf Schönheimer Institute of Biochemistry within Leipzig University’s Faculty of Medicine. Her lab has spent more than a decade focused on a still-underexplored family of proteins called adhesion G protein-coupled receptors (aGPCRs), a class of cell-surface receptors that act like biological antennas, translating physical forces and chemical signals from the outside world into instructions the cell can act on.
The lead author of the most recent bone study is Dr. Juliane Lehmann, also based at Leipzig’s Biochemistry Institute. Their work is embedded within Collaborative Research Centre 1423, a major scientific initiative dedicated to understanding the structural dynamics of GPCR activation and signaling. Leipzig is internationally recognized as one of the leading centers for this specialized area of molecular biology.
Importantly, the research has international reach. An earlier study by the Liebscher group conducted in collaboration with Shandong University in China had already shown that activating GPR133 with AP503 strengthens skeletal muscle. The new bone findings build naturally on that earlier discovery, painting an increasingly compelling picture of GPR133 as a master regulator of musculoskeletal health.
What Exactly Is GPR133, and Why Does It Matter?
GPR133 (also known as ADGRD1) is a receptor that sits on the surface of bone-forming cells called osteoblasts. Think of it as a sensory hub: it detects two distinct signals simultaneously, physical mechanical strain (the kind that comes from movement and exercise) and a molecular partner called PTK7, which is presented by neighboring cells.
When both signals arrive together, GPR133 fires. It triggers a cascade inside the cell: levels of cyclic AMP (cAMP) rise, which activates enzymes that ultimately switch on the β-catenin signaling pathway, a well-known driver of bone-building gene programs. The result is more osteoblast activity, more new bone formation, and, critically, less osteoclast activity (osteoclasts are the cells that break bone down). Bones become denser and stronger.
When the researchers genetically removed GPR133 from mice entirely, those animals developed thin, weak bones early in life, eerily similar to human osteoporosis. That finding alone validated GPR133 as a meaningful player in bone health. Human genome-wide association studies had previously linked GPR133 gene variants to differences in bone mineral density and even body height, but this was the first deep mechanistic look at exactly how and why.
Enter AP503: A Compound That Mimics the Body’s Own Signal
AP503 was identified through a computer-assisted screening process, essentially a sophisticated computational search designed to find small molecules capable of activating GPR133. It is classified as a selective receptor agonist, meaning it binds to GPR133 and mimics the natural activation that normally requires both mechanical force and PTK7 signaling.
When healthy mice received daily injections of AP503, their bone volume and strength increased measurably. When mice engineered to model postmenopausal osteoporosis (via ovariectomy, which removes estrogen) received the compound, the bone loss was significantly reversed, osteoblast counts improved and signs of bone resorption eased. Critically, when AP503 was combined with treadmill exercise, the effects were synergistic: the two together outperformed either intervention alone. That biological partnership between movement and a targeted molecule is exactly the kind of finding that excites longevity researchers.
As Dr. Lehmann noted, the newly demonstrated parallel strengthening of bone — building on their earlier muscle research — “highlights the great potential this receptor holds for medical applications in an aging population.”
What About Osteoarthritis?
While the primary focus of this research has been osteoporosis, the implications extend into osteoarthritis territory as well. Osteoarthritis involves the breakdown of cartilage in joints, often compounded by the weakening of the surrounding bone structure. As researchers continue to map GPR133’s role in skeletal tissue more broadly, its influence on bone quality at joint surfaces becomes a natural next question — and one the Leipzig team is actively pursuing in follow-up projects.
Where Are We on the Road to Human Treatment?
This is where excitement must be tempered with patience though not pessimism.
As of now, human clinical trials have not yet begun. The research is at the preclinical stage, conducted entirely in mouse models. Questions about long-term safety, potential off-target effects, and human pharmacokinetics (how the body processes the drug) remain open.
However, the Leipzig team is actively pursuing follow-up projects to explore AP503 in additional disease contexts and to deepen understanding of GPR133 across different tissues. Researchers also plan to study how individuals with different GPR133 genetic variants respond to the compound, a step toward precision medicine.
A realistic clinical timeline: additional preclinical safety studies over the next two to three years, followed by Phase 1 human trials (focused on safety and dosing) potentially by the late 2020s, with efficacy trials extending beyond that. Drug development is rarely fast — but the foundation being laid here is unusually solid, with strong mechanistic understanding, clear genetic human links, and a well-funded institutional framework behind it.
Why This Discovery Matters Especially for Women
Osteoporosis is not gender-neutral. In Germany alone, approximately six million people live with the condition, the majority of them women. Globally, postmenopausal women account for the largest share of osteoporosis cases, driven by the steep drop in estrogen that accelerates bone resorption. The fact that AP503 reversed bone loss in an ovariectomy mouse model, the gold-standard preclinical simulation of postmenopausal osteoporosis, is particularly meaningful.
Current treatments like bisphosphonates (which slow bone breakdown) and parathyroid hormone analogs (which stimulate formation) have real limitations: side effects, inconvenient delivery methods, and loss of efficacy over time. A receptor-targeted therapy that works with the body’s natural sensing machinery, and that pairs synergistically with exercise, could represent a genuinely different kind of tool.
The Bigger Picture
What makes this research feel different from many “promising mouse studies” is the layered quality of the science. The GPR133 story is grounded in human genetic data, validated through multiple mouse models, connected to real molecular pathways, and already linked to muscle health — suggesting a possible dual therapeutic benefit for the increasingly recognized syndrome of combined muscle and bone loss in aging.
The body, it turns out, may already know how to build strong bones. It just needs the right signal. Scientists in Leipzig have found a way to send it.
Source: “The mechanosensitive adhesion G protein-coupled receptor 133 (GPR133/ADGRD1) enhances bone formation”
Signal Transduction and Targeted Therapy (Nature Publishing Group), 2025
Leave a comment