There’s a quietly growing conversation in nootropic and neurological research circles about a short-chain peptide called Semax and for good reason. Originally developed behind the Iron Curtain, Semax has since emerged as one of the more compelling neuropeptides studied for its cognitive and neuroprotective properties. Here’s what the science says.
Semax is a Nasal-Spray
Origins: From Soviet Lab to Modern Research
Semax was developed in the 1980s at the Institute of Molecular Genetics of the Russian Academy of Sciences. Researchers were investigating adrenocorticotropic hormone (ACTH) and its derivatives when they identified a short fragment, ACTH(4–7) , that retained neurological activity without the hormonal side effects of the parent molecule. They then added a C-terminal proline-glycine-proline (Pro-Gly-Pro) sequence to stabilize it and enhance its CNS penetration, and Semax was born.
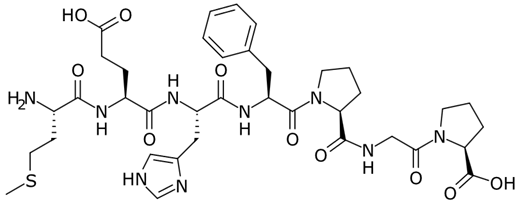
Semax peptide structure
The full sequence is Met-Glu-His-Phe-Pro-Gly-Pro. It was approved in Russia in 1995 for clinical use in conditions such as stroke recovery and transient ischemic attacks. It remains in use there today, typically administered intranasally.
What Is Semax Used For?
Clinically, Semax has been used in Russia and parts of Eastern Europe for stroke rehabilitation, cognitive impairment, and optic nerve disorders. In research and off-label contexts, it has attracted interest for memory enhancement, attention, anxiety reduction, and neuroprotection after brain injury. Semax [9] has undergone extensive study in Russia and is on the Russian List of Vital & Essential Drugs approved by the Russian Federation government on 7 December 2011. Medical uses for Semax include treatment of stroke, transient ischemic attack, memory and cognitive disorders, peptic ulcers, optic nerve disease, and to boost the immune system.
Its intranasal delivery route is a notable feature; it allows the peptide to bypass the blood-brain barrier through olfactory pathways, delivering it more directly to the central nervous system than an oral or peripheral route would allow.
Mechanism: Receptor Binding and Downstream Cascades
Semax is not a simple receptor agonist. Its mechanism is multifaceted, but the most well-characterized pathway involves the upregulation of brain-derived neurotrophic factor (BDNF) and its primary receptor, TrkB (tropomyosin receptor kinase B).
When Semax is administered, it stimulates BDNF gene expression in the hippocampus and frontal cortex, regions critical for memory consolidation and executive function. BDNF binding to TrkB activates several downstream signaling cascades, most notably:
- MAPK/ERK pathway — involved in synaptic plasticity and long-term memory formation
- PI3K/Akt pathway — promotes neuronal survival and inhibits apoptosis
- PLCγ pathway — modulates intracellular calcium and synaptic transmission
Additionally, Semax influences the dopaminergic and serotonergic systems and has demonstrated anti-inflammatory effects by modulating gene expression in immune-related neural pathways. Accordingly, it has been found to produce antidepressant-like and anxiolytic-like effects, attenuate the behavioral effects of exposure to chronic stress, and potentiate the locomotor activity produced by D-amphetamine. As such, it has been suggested that Semax may be effective in the treatment of depression.
Though the exact mechanism of action of Semax is unclear, there is evidence that it may act through melanocortin receptors. Specifically, there is a report of Semax competitively antagonizing the action of α-melanocyte-stimulating hormone (α-MSH) at the MC4 and MC5 receptors in both in vitro and in vivo experimental conditions, indicating that it may act as an antagonist or partial agonist of these receptors. (&alpha-MSH acts as a full agonist of all five melanocortin receptors). Semax did not antagonize α-MSH at the MC3 receptor, though this receptor could still be a target of the drug. As for the MC1 and MC2 receptors, they were not assayed.

What the Studies Say
Human and animal research on Semax is primarily Russian in origin, which limits widespread peer review access, but several meaningful findings have emerged.
A 2001 study published in the Bulletin of Experimental Biology and Medicine demonstrated that Semax significantly improved cognitive outcomes in patients recovering from ischemic stroke. Patients showed improved attention, memory, and daily functioning versus controls.
Animal studies have shown Semax protects neurons from hypoxic and excitotoxic damage, reduces infarct size in stroke models, and accelerates learning in maze-based behavioral tasks. A 2011 study in rats showed Semax administration upregulated BDNF mRNA in the hippocampus within hours of dosing, providing a plausible molecular basis for its cognitive effects.
More recent transcriptomic analyses, notably work by Medvedeva et al., mapped over 800 gene expression changes in rat brain tissue following Semax administration, many related to immune regulation, neurogenesis, and synaptic function. This suggests the peptide’s effects are broad and system-wide rather than narrowly targeted.
That said, large-scale randomized controlled trials in Western research populations remain sparse. The existing evidence is promising but preliminary by current standards.
Contraindications and Adverse Events
Semax has a generally favorable safety profile in published literature. Reported adverse events are mild and typically include nasal irritation at the site of intranasal administration, occasional mild anxiety or irritability, and in some cases, temporary fatigue.
More significant cautions include its use in individuals with a history of seizures, as pro-BDNF activity has theoretical potential to lower seizure threshold in susceptible individuals. It should also be used with caution in those with psychiatric conditions, given its influence on dopaminergic signaling.
Pregnant and breastfeeding individuals should avoid Semax due to absence of safety data. There are no established drug interaction profiles in human literature, which is itself a limitation worth noting.
The Bottom Line
Semax is a genuinely interesting peptide with a plausible mechanism, supportive animal data, and encouraging early human studies, particularly in neurological rehabilitation. It warrants serious scientific attention and larger, independently replicated trials. Until that evidence matures, it sits in a familiar place in modern neuroscience: promising, but not yet fully proven.
This post is for educational purposes only and does not constitute medical advice. Consult a qualified healthcare provider before considering any peptide therapy.
Leave a comment