In the landscape of herbal substances that blur the line between botanical remedy and controlled drug, few have generated as much scientific curiosity, political controversy, and passionate public debate as kratom. Whether you’ve heard of it from a chronic pain forum, a biohacking podcast, or a news headline about federal regulation, there’s more to this Southeast Asian leaf than most people realize, and the story of how it nearly became federally banned tells us something important about the power of informed citizens to shape public health policy.
From Jungle Leaf to American Shelves

Kratom comes from Mitragyna speciosa, a tropical tree in the coffee family (Rubiaceae) native to Southeast Asia — particularly Thailand, Malaysia, Indonesia, and Papua New Guinea. For centuries, laborers in these regions chewed the leaves to combat fatigue, manage pain, and increase productivity in grueling agricultural conditions. It was folk medicine in the truest sense: low-tech, locally sourced, and passed down through generations.
Today, kratom arrives in American homes as a powder, capsule, or extract — a far cry from fresh-chewed leaves, but the same basic chemistry underneath.
The Chemistry: Two Alkaloids Doing Heavy Lifting
The pharmacological story of kratom centers on its two primary bioactive alkaloids: mitragynine and 7-hydroxymitragynine. Together, these compounds account for the vast majority of kratom’s effects. Mitragynine makes up roughly 60–70% of the total alkaloid content, while 7-hydroxymitragynine is present in much smaller quantities — but is considerably more potent.
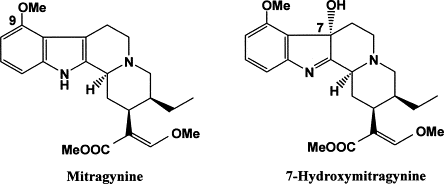
Both compounds are indole-based alkaloids with structural features that allow them to interact with opioid receptors in the human brain and body. Their chemical architecture gives them the ability to fit into receptor binding pockets in ways that produce opioid-like effects, though they are structurally distinct from classical opioids like morphine or oxycodone.
How It Works: Mu Receptors, G-Proteins, and a Key Divergence
To understand what kratom does in the body, you need a quick primer on opioid receptor pharmacology. The mu-opioid receptor (MOR) is the primary target for most pain-relieving and euphoria-producing opioids. When a drug like morphine binds to the MOR, it triggers two main downstream pathways: one involving G-proteins and another involving molecules called beta-arrestins.
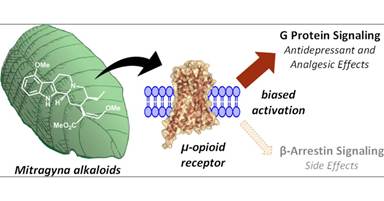
The G-protein pathway is largely responsible for the desirable effects — analgesia, sedation, and euphoria. The beta-arrestin pathway, by contrast, is associated with many of the problematic consequences of opioid use: respiratory depression, tolerance, and constipation.
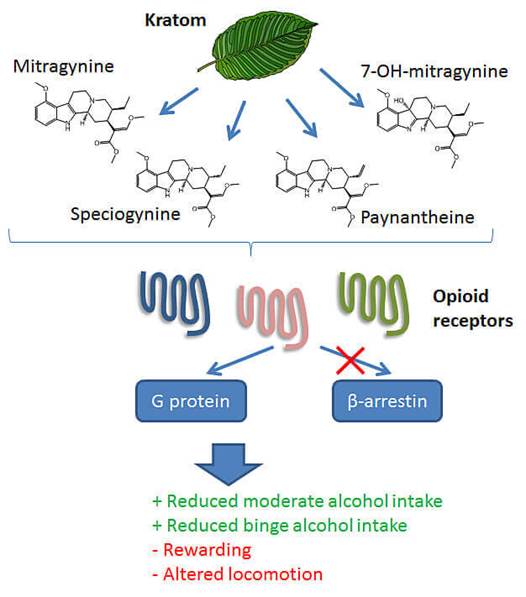
Here is where kratom’s alkaloids get genuinely interesting from a pharmacological standpoint. Both mitragynine and 7-hydroxymitragynine are partial agonists at the mu-opioid receptor, activating it through G-protein signaling — but critically, they appear to show biased agonism, meaning they activate the G-protein pathway while having relatively little activity at the beta-arrestin pathway. This property, known as “G-protein bias,” is the same pharmacological quality that researchers have been chasing for years in the development of “safer” opioid painkillers. The theory is that a G-protein biased opioid agonist could provide pain relief and mood elevation with a reduced risk of the dangerous side effects tied to beta-arrestin signaling, particularly respiratory depression — the mechanism by which opioid overdoses become fatal.
This does not make kratom safe — it makes it scientifically interesting, which is a different thing entirely.
Why People Use It
Despite having no FDA-approved medical indication, an estimated 1 to 15 million Americans use kratom regularly, according to various survey estimates. They’re reaching for it for a cluster of overlapping reasons:
Pain management is the most commonly cited use. Many users are chronic pain patients who feel underserved by conventional medicine, particularly in the context of prescription opioid restrictions following the opioid crisis. For them, kratom fills a gap — imperfectly and unofficially.
Recreational effects and euphoria are also part of the picture, particularly at higher doses. At lower doses, kratom tends to produce stimulant-like effects: increased alertness and energy. At higher doses, it shifts toward sedation and euphoria, more typical of opioid-class substances.
Anxiety and stress relief round out the picture. Many users describe kratom as calming without the cognitive fog associated with benzodiazepines, making it appealing for stress management.
Safety Profile and the Research Gap
Here’s the honest assessment: the safety profile of kratom is poorly characterized. Clinical research is sparse, long-term human data is limited, and most of what we know comes from case reports, surveys, and animal studies. The FDA has attributed a number of deaths to kratom, though critics note that most of these cases involved polydrug use, making causation difficult to establish.
Known risks include nausea, liver toxicity (in rare cases), dependence, and withdrawal symptoms. The absence of robust clinical trials means neither a full harm profile nor a clear therapeutic window has been established.
The DEA’s Attempted Scheduling — and a Remarkable Public Pushback
In August 2016, the DEA made an extraordinary announcement: it planned to place kratom’s primary alkaloids into Schedule I of the Controlled Substances Act — the most restrictive category, reserved for drugs with “no accepted medical use” and “high abuse potential,” alongside heroin and LSD. The agency invoked its emergency scheduling authority, intending to bypass the usual public comment process.
The public response was swift and organized. Tens of thousands of citizens submitted comments. Patients, researchers, and advocacy groups flooded congressional offices with calls and letters. A bipartisan group of 51 members of Congress signed a letter asking the DEA to withdraw the emergency scheduling action and allow for scientific review. The American Kratom Association organized nationally.
In an unusual reversal, the DEA withdrew its intent to emergency schedule kratom in October 2016, citing the unprecedented public response and agreeing to an open comment period. The FDA has since issued warnings and pursued enforcement actions against kratom products, but as of this writing, kratom remains legal at the federal level although several states have banned it individually.
This episode is a compelling case study in civic engagement. A well-informed, organized public, armed with scientific arguments and personal testimony, reversed a major regulatory agency’s plans. Whatever your views on kratom, the policy episode is a reminder that public participation in regulatory processes is not theater; it can actually work.
Dependency, Addiction, and the Opioid Withdrawal Question
Kratom’s partial agonist activity at mu-opioid receptors means it does carry dependence and addiction potential. Regular users can develop tolerance and experience withdrawal symptoms upon cessation, including muscle aches, insomnia, irritability, and anxiety. These symptoms resemble, though are generally considered milder than, classic opioid withdrawal.
Perhaps most intriguingly, kratom is used by some individuals as a self-managed tool for opioid withdrawal and harm reduction essentially stepping down from more potent opioids using kratom as a bridge. Anecdotal reports describe meaningful symptom relief. However, this practice has not been validated in clinical trials, carries its own dependency risks, and is not endorsed by medical authorities. Replacing one opioid-acting substance with another is a strategy that requires careful medical supervision, not a DIY solution.
The Bottom Line
Kratom sits in a genuinely complicated space — scientifically fascinating, pharmacologically plausible in some of its claimed mechanisms, politically volatile, and practically under-studied. Its G-protein biased mechanism of action is legitimately interesting to researchers. Its role in the lives of pain patients is real and often deeply felt. Its risks are real and not fully mapped. And its near-scheduling and subsequent reversal is a rare, instructive example of democratic engagement shaping drug policy.
If you’re a patient, a biohacker, or simply someone curious about the frontier where ethnobotany meets neuropharmacology, kratom deserves your critical attention — not uncritical enthusiasm, and not reflexive dismissal. What it deserves most, frankly, is the rigorous clinical research that has so far been lacking. Until that exists, the conversation will remain exactly where it is: fascinating, unresolved, and urgently worth having.
This article is for informational purposes only and does not constitute medical advice. If you are managing pain, opioid use disorder, or considering kratom, consult a qualified healthcare provider.
Leave a comment