From Wound Healing to Systemic Rejuvenation: Understanding the Science Behind the “Fountain of Youth” Peptide
In the landscape of anti-aging interventions, few compounds have generated as much excitement and scientific validation as GHK-Cu. This isn’t just another peptide promising youthful skin; it’s a naturally occurring tripeptide-copper complex that appears to fundamentally reprogram how cells express their genetic information. If the Wolverine Stack is about accelerated healing, GHK-Cu is about cellular rejuvenation at the most fundamental level.
The Discovery: When Old Cells Act Young
The story of GHK-Cu begins in 1973 at the University of California, San Francisco, where Dr. Loren Pickart was investigating a puzzling observation: liver cells from elderly patients functioned dramatically better when cultured in blood plasma from younger donors. Something in young blood was making old cells behave like young cells.
Through painstaking fractionation and isolation techniques, Pickart identified the responsible factor: a small tripeptide consisting of just three amino acids, Glycine-Histidine-Lysine (GHK) bound to a copper ion (Cu²⁺). When he measured GHK-Cu levels across different age groups, he found a striking pattern. At age twenty, the average plasma concentration hovered around 200 nanograms per milliliter. By age sixty, that concentration had dropped to approximately 80 nanograms per milliliter, a decline of about sixty percent over four decades.
This wasn’t just correlation. When Pickart added GHK-Cu back to aged cells, they demonstrated restored function, producing proteins and responding to signals like younger cells. The implications were profound: aging might not be simply about accumulated damage but also about the loss of specific signaling molecules that maintain youthful cellular behavior.
Molecular Structure: The Copper Connection
GHK-Cu’s structure is deceptively simple but functionally sophisticated. The tripeptide sequence creates a specific three-dimensional shape that forms a “binding pocket” with extraordinarily high affinity for copper ions, specifically Cu²⁺ (cupric copper). The dissociation constant is in the picomolar range, meaning once GHK binds copper, it holds on extremely tightly.
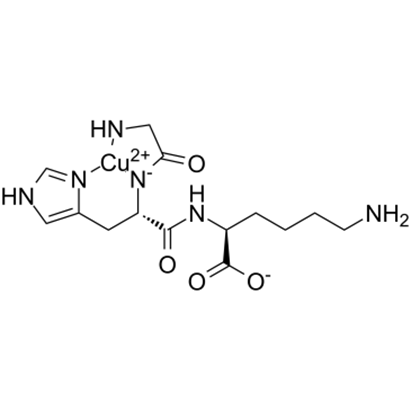
Copper isn’t just a passive passenger in this molecular complex; it’s integral to GHK-Cu’s biological activity. Copper can cycle between Cu²⁺ and Cu⁺ states, making it a critical cofactor in redox reactions, the electron transfer reactions that underlie most cellular energy processes. Copper is required for several critical enzymes that govern tissue health and repair. Superoxide Dismutase, one of the body’s primary antioxidant enzymes, requires copper to neutralize superoxide radicals. Lysyl Oxidase, the enzyme that cross-links collagen and elastin fibers to give tissue its structural integrity, is copper-dependent. Even Tyrosinase, involved in melanin production and wound healing, needs copper to function.
Beyond its role as an enzyme cofactor, copper ions themselves can act as signaling molecules, influencing gene expression through copper-responsive transcription factors. The GHK peptide acts as a copper chaperone, safely delivering copper to cells that need it while preventing toxic accumulation. Free copper is highly reactive and can generate harmful free radicals through Fenton chemistry; GHK-bound copper is stable and biologically directed.
Cellular Mechanisms: Epigenetic Reprogramming
GHK-Cu’s effects operate at multiple levels, but its most profound impact is on gene expression, the process by which cells “read” their DNA to produce proteins. This is where the science becomes truly fascinating.
In 2012, researchers used the Connectivity Map database, a comprehensive collection of gene expression profiles from cells treated with various compounds to analyze GHK-Cu’s effects. The results were staggering. GHK-Cu significantly altered the expression of over four thousand genes, systematically shifting them toward patterns seen in younger tissue. Specifically, approximately eighteen hundred genes were upregulated while over two thousand genes were downregulated.
This wasn’t random modulation. The changes showed clear, biologically meaningful patterns. The upregulated genes were involved in tissue repair and remodeling, antioxidant defense, anti-inflammatory pathways, DNA repair mechanisms, and stem cell mobilization. Meanwhile, the downregulated genes included pro-inflammatory cytokines like IL-1, IL-6, and TNF-α; matrix metalloproteinases that degrade collagen when overexpressed; genes associated with fibrosis and scar formation; and even certain cancer-promoting oncogenes, including those associated with metastasis.
This represents epigenetic modulation, changing how genes are expressed without altering the DNA sequence itself. Think of your genome as a massive library containing every instruction your cells might need. GHK-Cu acts like a master librarian, determining which books get pulled from the shelves and which remain closed. The shift isn’t subtle; it’s a systematic reorganization toward a more youthful expression pattern.
Continue for part 2 …
Leave a comment